Explanation of Intermolecular Forces:
There are two intermolecular forces that act upon the chemical compound of ammonium nitrate:
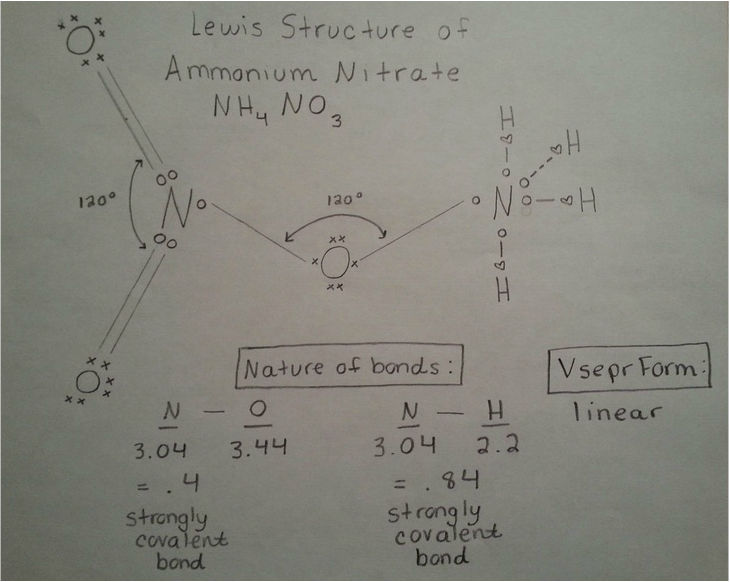
One of the forces is hydrogen-bonding, which is when the hydrogen (H) of one molecule is attracted to the oxygen (O), nitrogen (N), or fluorine (F) of another molecule. In this chemical compound, the H in NH4 form hydrogen bonds with the O in NO3.
In addition, dispersion, which is the force that acts on every and any two adjacent molecules, acts on ammonium nitrate.
One of the forces is hydrogen-bonding, which is when the hydrogen (H) of one molecule is attracted to the oxygen (O), nitrogen (N), or fluorine (F) of another molecule. In this chemical compound, the H in NH4 form hydrogen bonds with the O in NO3.
In addition, dispersion, which is the force that acts on every and any two adjacent molecules, acts on ammonium nitrate.