Basic Facts About Ammonium Nitrate:
Ammonium nitrate is a fascinating chemical compound that is capable of creating both common fertilizers and deadly explosions. It is expressed through the chemical formula NH4NO3. At room temperature, ammonium nitrate is in the form of white, odorless, solid pellets. Its melting point is 169.6 °C, which is equivalent to 337.3 °F. Its boiling point is 210 °C, which is equivalent 410 °F. It is highly soluble in water at 1920 g/L. Its density is 1.72 g/cm3.
Ammonium Nitrate as an Explosive:
Ammonium nitrate is a very hazardous compound that can be harmful to humans because of its chemical components and its capability of explosion. Although it is a relatively stable chemical compound, the government requires it to be handled with special care because of its dangerous qualities. Ammonium nitrate does not burn when ignited with flame, but instead melts and decomposes. However, it is very sensitive to heat, and a high amount of activation energy and a significant amount of heat will cause an ammonium nitrate explosion. If an explosion does occur, it is likely in the presence significantly high temperatures.
A combination of factors can result in an ammonium nitrate explosion. These factors include being exposed to a great deal of activation energy and being enclosed in a tight space at a high temperature. Explosions due to the confinement of ammonium nitrate in storage or after production are influenced by insufficient flow throughout the space, poor maintenance of the space, and contamination. If stored or used improperly under certain conditions, ammonium nitrate explosions can occur with extremely dangerous and destructive consequences.
Because ammonium nitrate is a powerful oxidizing agent, it can take away electrons from other reactants when involved with a redox chemical reaction. It is because of this unique quality that ammonium nitrate is an explosive and is incorporated into substances with combustibles such as TNT for the creation of explosives.
When ammonium nitrate fertilizer is combined with fuel in ideal proportions, bombs can be created and explosions can occur. During a fertilizer bomb explosion, the detonator is the first to explode, releasing a denotation wave. The energy from this wave triggers the ammonium nitrate to vaporize, in which the solid immediately becomes a gas. During this time, the ammonium and nitrate molecules separate and decompose, instantly resulting in a substantial quantity of oxygen gas. This gas essentially causes the explosion. The fuel catches on fire because of a combination of the formation of the oxygen and the denotation wave energy, resulting in the combustion of the fuel and the creation of more gas. A great deal of gas is generated in brevity, resulting in pressure waves, which causes the most damage in an explosion.
A combination of factors can result in an ammonium nitrate explosion. These factors include being exposed to a great deal of activation energy and being enclosed in a tight space at a high temperature. Explosions due to the confinement of ammonium nitrate in storage or after production are influenced by insufficient flow throughout the space, poor maintenance of the space, and contamination. If stored or used improperly under certain conditions, ammonium nitrate explosions can occur with extremely dangerous and destructive consequences.
Because ammonium nitrate is a powerful oxidizing agent, it can take away electrons from other reactants when involved with a redox chemical reaction. It is because of this unique quality that ammonium nitrate is an explosive and is incorporated into substances with combustibles such as TNT for the creation of explosives.
When ammonium nitrate fertilizer is combined with fuel in ideal proportions, bombs can be created and explosions can occur. During a fertilizer bomb explosion, the detonator is the first to explode, releasing a denotation wave. The energy from this wave triggers the ammonium nitrate to vaporize, in which the solid immediately becomes a gas. During this time, the ammonium and nitrate molecules separate and decompose, instantly resulting in a substantial quantity of oxygen gas. This gas essentially causes the explosion. The fuel catches on fire because of a combination of the formation of the oxygen and the denotation wave energy, resulting in the combustion of the fuel and the creation of more gas. A great deal of gas is generated in brevity, resulting in pressure waves, which causes the most damage in an explosion.
|
|
|
Production/Manufacturing of Ammonium Nitrate:
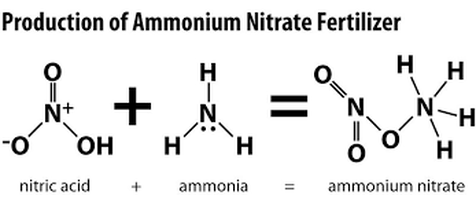
Ammonium nitrate is produced from the manmade, induced exothermic reaction between ammonia and nitric acid. During the production of the chemical compound, ammonia gas essentially reacts with nitric acid, creating a great deal of heat and a concentrate solution. Some of the processes by which ammonium nitrate is produced include the “Haber-Bosch” process, the “Odda” process, and a nitrophosphate process. Steps involved in the production of ammonium nitrate include neutralization, evaporation, prilling and granulation, cooling, and conditioning. It is through prilling and granulation that ammonium nitrate becomes a solid.
There are several forms and functions of the ammonium nitrate, and because of this, there are a variety of methods/processes used to produce it. Despite the differences between the ways it is manufactured, the essence of how it is produced is the same in that ammonia is reacted with nitric acid. For example, in the prilling method/stage of manufacturing ammonium nitrate, low-density prills are utilized when producing the chemical compound for industrial or explosive use. High-density prills are utilized when producing the chemical compound for fertilizer. Ammonium nitrate in granular form, in which it is in the form of very small particles or grains, is produced by continuously spraying tiny granules with a specific concentrated solution within a rotating drum. No matter what method is utilized, the production of this chemical compound is meticulous and requires serious diligence and caution.
There are several forms and functions of the ammonium nitrate, and because of this, there are a variety of methods/processes used to produce it. Despite the differences between the ways it is manufactured, the essence of how it is produced is the same in that ammonia is reacted with nitric acid. For example, in the prilling method/stage of manufacturing ammonium nitrate, low-density prills are utilized when producing the chemical compound for industrial or explosive use. High-density prills are utilized when producing the chemical compound for fertilizer. Ammonium nitrate in granular form, in which it is in the form of very small particles or grains, is produced by continuously spraying tiny granules with a specific concentrated solution within a rotating drum. No matter what method is utilized, the production of this chemical compound is meticulous and requires serious diligence and caution.
Storage of Ammonium Nitrate:
The proper storage of ammonium nitrate is extremely important because of its potentially hazardous qualities. During the storage of ammonium nitrate in solution form, the gas state of ammonia should be frequently added in small amounts in order to sustain the ideal pH and account for any ammonia lost while in storage. In addition, the confinement of ammonium nitrate should be limited greatly. Proper ventilation, maintenance, and contamination control must be addressed, as inappropriate confinement can increase the risk of explosions. Ammonium nitrate is hygroscopic, meaning it can easily collect water molecules from its surrounding environment. Because of this, it cannot be exposed to excessive humidity, as any water absorbed by the compound can influence its explosive function. The careful storage of this chemical compound is a necessity and must be taken seriously.
Other Functions of Ammonium Nitrate:
Ammonium nitrate is often used in fertilizers because it provides plants with a form of nitrogen that they can use in their natural chemical processes. It can function directly or indirectly in fertilizers. The nitrate part of ammonium nitrate helps water in the soil travel to the roots for immediate use by the plant. Ammonium nitrate was the first solid nitrogen fertilizer to be manufactured on a large scale. This chemical compound, when incorporated into fertilizer, is utilized for its fairly high nutrient content and good balance of both ammonia and nitrate. Ammonium nitrate makes an excellent fertilizer, with significant economic and environmental benefits from its use. When incorporated into a fertilizer, ammonium nitrate is no longer capable of exploding because it is combined with several other chemicals such as ammonium sulfate or urea.
In addition, it can be used in the production of bombs because of its explosive qualities and capabilities.
In addition, it can be used in the production of bombs because of its explosive qualities and capabilities.
Summary of Hazards of Ammonium Nitrate Use and Exposure:
As previously explained, ammonium nitrate is a hazardous chemical compound because of its explosive capabilities. Those who are exposed to ammonium nitrate by manufacturing or using it are at risk of these explosions, emphasizing the importance of proper handling and storage. In addition to the risk of being injured or killed in an explosion, those handling and working with this chemical compound are in danger of respiratory tract irritation, caused by the inhalation of high concentrates of ammonium nitrate dust. The numerous risks of ammonium nitrate exposure highlight the necessity of responsible and careful use.